|
As the position of the particle becomes more precise when the slit is narrowed, the direction, or therefore the momentum, of the particle becomes less known as seen by a wider horizontal distribution of the light.ġ.) The Heisenberg Uncertainty Principle discredits the aspect of the Bohr atom model that an electron is constrained to a one-dimensional orbit of a fixed radius around the nucleus.Ģ. Here is a video that demonstrates particles of light passing through a slit and as the slit becomes smaller, the final possible array of directions of the particles becomes wider. Therefore, the momentum is unknown, but the initial position of the particle is known. He had light passing through a slit, which causes an uncertainty of momentum because the light behaves like a particle and a wave as it passes through the slit. Einstein created a slit experiment to try and disprove the Uncertainty Principle. Several scientists have debated the Uncertainty Principle, including Einstein. 
Therefore, there is no way to find both the position and momentum of a particle simultaneously. Conversely, if we want a more precise momentum, we would add less wavelengths to the "wave packet" and then the position would become more uncertain. The more waves that are combined in the "wave packet", the more precise the position of the particle becomes and the more uncertain the momentum becomes because more wavelengths of varying momenta are added. An accumulation of waves of varying wavelengths can be combined to create an average wavelength through an interference pattern: this average wavelength is called the "wave packet". A "wave packet" can be used to demonstrate how either the momentum or position of a particle can be precisely calculated, but not both of them simultaneously. However, in quantum mechanics, the wave-particle duality of electrons does not allow us to accurately calculate both the momentum and position because the wave is not in one exact location but is spread out over space. It is hard for most people to accept the uncertainty principle, because in classical physics the velocity and position of an object can be calculated with certainty and accuracy. Understanding the Uncertainty Principle through Wave Packets and the Slit Experiment Neither uncertainty can become small without the other becoming large. It is impossible to measure position x and momentum p simultaneously with uncertainties x and p that multiply to be less than h 4 h 4. However, the more accurately momentum is known the less accurately position is known. This is known as the Heisenberg uncertainty principle. 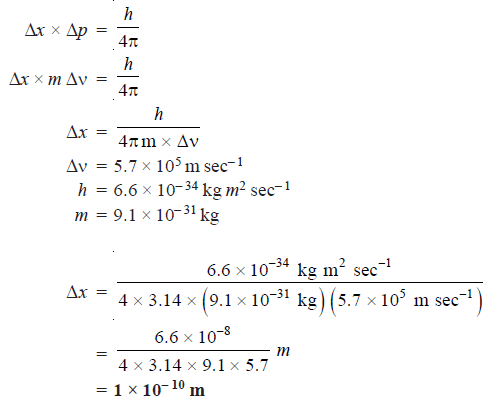
Mathematically, this occurs because the smaller Δx becomes, the larger Δp must become in order to satisfy the inequality. 
What this equation reveals is that the more accurately a particle’s position is known, or the smaller Δx is, the less accurately the momentum of the particle Δp is known.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
